Cochrane Kidney and Transplant (CKT) develop and maintain a database known as the Cochrane Kidney and Transplant Register of Studies.
 It is a database of randomised controlled trials in kidney disease that reflects the scope of our Group. It has been developed primarily to support authors writing systematic reviews, although it is also a unique and comprehensive resource for the nephrology community, providing support for the development of quality evidence-based practice and research by kidney physicians, nurses, researchers, policy makers, other health-related professionals, and consumers.
It is a database of randomised controlled trials in kidney disease that reflects the scope of our Group. It has been developed primarily to support authors writing systematic reviews, although it is also a unique and comprehensive resource for the nephrology community, providing support for the development of quality evidence-based practice and research by kidney physicians, nurses, researchers, policy makers, other health-related professionals, and consumers.
The database is study-based i.e. all reports or publications of a study (or trial) are linked together under a unique Study name (example of a study record in the database). It is a relational database containing review information and bibliographic and study details, where the STUDY is the unit of data. The decision to develop a study-based database was based on the following assumptions that:
- the identification of duplicate publications is vital to producing an accurate systematic review. If multiple reports of a single trial are included as individual trials i.e. the trial participants are counted multiple times, then the effect of a particular intervention may be over or under estimated, leading to wrong conclusions of its efficacy. Unless all reports or publications from a study were linked, the possibility exists of including data from the same trial more than once in a systematic review;
- a study-based register would be able to reduce the workload of authors in the screening process
The database is currently maintained in MeerKat, study-based software based on MS-Access. The database:
- contains bibliographic and study details and review information where the study is the unit of data
- links multiple reports to a single study
- links studies to reviews
- tracks the progress of reviews by linking studies sent to and used by authors
The database is populated from a variety of sources: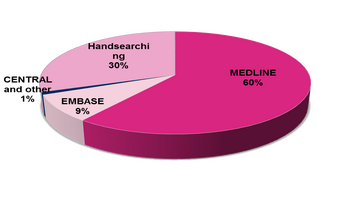
- Monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL)
- Weekly searches of MEDLINE OVID SP
- Searching of the current year of EMBASE OVID SP for each review
- Searches of kidney and transplant journals and the proceedings of major kidney and transplant conferences
- Weekly current awareness alerts for selected kidney and transplant & general journals
- Searches of the International Clinical Trials Register (ICTRP) Search Portal and ClinicalTrials.gov for ongoing studies
Strategies for the identification of randomised controlled trial for the CKT Register of Studies:
- Search strategies for electronic databases
- Searching other resources - journals and conference proceedings
All reports are screened for relevance in terms of the scope of Cochrane Kidney and Transplant & study design (i.e randomised controlled trial (RCT) or quasi-randomised controlled trial (CCT)). The full-text of all potential reports is obtained.
Each report is checked against MeerKat to see if the report is part of a study that already exists. If so, the report is added to that study in MeerKat.
If the report is a new study, a new study is created and the reference added to that study. Individual reports can be linked to more than 1 study.
Studies and reports are coded with interventions, health care conditions and a review number (i.e. a review that is registered or published with CKT) to which the study may be relevant. A potential review title may also be added. Study registration IDs (e.g. NCT001123456) are also obtained and added where available.
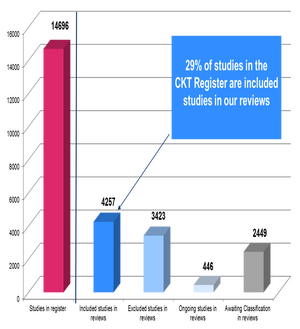
The CKT Register of Studies contains 28045 reports of 14969 studies.
As of October 2020, 29% (n=4257) of the studies in the CKT Register of Studies (n=14696) are Included Studies in CKT systematic reviews. Another 18% (n=2490) of studies are Awaiting Classification in CKT systematic reviews currently in production.
Extra details about the content of the CKT Register of Studies
